Casefinding Introduction
On this page...
- Casefinding Background
- Casefinding Purpose and Importance
- Casefinding and Legislation
- Casefinding Lists
- Reportable List
Casefinding Background
Casefinding is a system for locating every patient - inpatient or outpatient, who is diagnosed and/or treated with a reportable diagnosis. Casefinding is like casting a net far and wide to capture all of the reportable cancer cases. This is to ensure that all cancers are included in the hospital cancer registry database or a population-based cancer registry database.
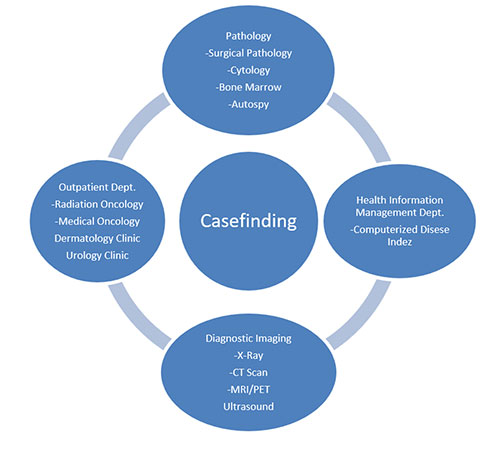
Casefinding Purpose and Importance
Casefinding is an important and necessary part of the cancer registry and national cancer surveillance. A system to monitor prospective cases must be in place in different areas of an institution. The completeness of casefinding must be monitored for quality control purposes.
In the casefinding process, a tracking system is kept so that the status of casefinding can be ascertained at any time. Most cancer registries are automated and have a software generated suspense list. A suspense list includes potentially reportable cancer cases identified by ICD-10-CM codes, International Classification of Diseases, Tenth Revision, Clinical Modification (U.S. only); which must be reviewed to determine if a case is reportable. All cancer registries must perform casefinding, including hospital-specific and central or population-based registries. Although these cancer registries may use different source documents, the procedures involved in casefinding cycles are similar.
Most national standard setting cancer registries in the United States such as the Surveillance, Epidemiology, and End Results (SEER) Program and the National Program of Cancer Registries (NPCR) only require malignant ICD-O-3, International Classification of Diseases for Oncology, Third Edition behavior codes 2 and 3 cases to be included in the cancer registry. However, starting in 2004, benign and borderline brain and central nervous system (CNS) cases are required to be reported in the United States. In addition, some hospitals and central registries may require borderline ovarian tumors, carcinoid tumors of the appendix, and squamous cell carcinoma of the skin that they may deem pertinent to conduct research.
Casefinding and Legislation
Authority for a population based cancer registry to collect data on cancer incidence is established through legislation for cancer reporting. This is determined by individual state or provincial/territorial health authorities.
Legislation or statute refers to a form of law enacted by a state, provincial/territorial legislature, Congress, or Parliament. Regulation or rule refers to a form of law created by administrative agencies of a government.
Legislative authority SHOULD include specific components that relate to central registry development and function, as well as specific directives for the publication of regulations detailing these components. Often, authority is granted to the jurisdiction’s health department, which, in turn, may delegate authority to another agency. In other instances, authority is granted directly to another agency, such as a university or foundation.
Casefinding Lists
A casefinding list is not the same as a reportable list. Casefinding lists are intended for searching a variety of sources as to not miss any reportable solid tumor, hematopoietic, or benign brain cases due to the omission of the word cancer.
For hematopoietic cases be sure to review the notes section in the Heme manual when screening for hematopoietic cases.
To avoid under-reporting of hematopoietic cases, follow back with the physician to confirm the diagnosis whenever possible.
[Refer to https://seer.cancer.gov/tools/casefinding/fy2020-casefindinglist-icd10cm.pdf for the current and previous casefinding list]Reportable List
- The reportable list https://seer.cancer.gov/manuals/2018/SPCSM_2018_maindoc.pdf (pgs. 6-8) should be posted in the cancer registry's Policy and Procedure Manual.
- The criteria for eligible cases in a cancer registry depend upon the governing agencies of the cancer registry. Along with state-specific reportable cases, cancer registries participating in the Approvals Program of the Commission on Cancer (CoC) of the American College of Surgeons (ACoS
) must use the reportable list defined by the CoC. https://www.facs.org/~/media/files/quality%20programs/cancer/ncdb/store_manual_2018.ashx
(p. 15)
- Exceptions to reporting requirements (Do not report)
- Primary skin cancers with histology (cell type) codes 8000-8110
- Carcinoma in situ of cervix, cervical intraepithelial neoplasia (CIN III) of the cervix
- Prostatic intraepithelial neoplasia (PIN III)
In addition, all healthcare facilities are required to submit cancer cases to their respective state cancer registry. Facilities should be familiar with their state cancer registry reporting requirements including the state cancer reporting manual which includes a reportable list. The reportable list of the state cancer registry should clearly state the type of cases that are in addition to those required by the CoC. Cases required by the CoC can be found in the STORE Manual, Case Eligibility Section. This ensures that cases that may otherwise be misclassified are reviewed during casefinding and reported to the state and national cancer registry. State cancer registry reportable list can be found on the individual state’s website (or contact your state cancer registry directly).
Coding Tip: It is the responsibility of the reporting facility to become familiar with the most current version of their respective state cancer registry manual and reporting requirements.
Casefinding lists vs Reportable list
- Refer to SEER Program Coding and Staging Manual 2018, Reportability Section
Reportable cases
- Refer to STORE Manual 2018, Case Eligibility Section
Non-reportable cases
- Refer to STORE Manual 2018, Case Eligibility Section
Updated: December 12, 2023
Suggested Citation
SEER Training Modules: Casefinding Introduction. U.S. National Institutes of Health, National Cancer Institute. Cited 10 June 2026. Available from: https://training.seer.cancer.gov.




